The density of water in kg/m3 has numerous applications in various fields, including: Applications of Density of Water in kg/m3: However, the change in density with pressure is relatively small and is typically only significant at very high pressures. This is because as the pressure on water increases, the water molecules become closer together, resulting in a higher mass per unit volume, or density. The density of water in kg/m3 also changes with pressure, with the density increasing as the pressure increases. The density of water in kg/m3 at different temperatures can be expressed as follows: Temperature (☌)ĭensity of Water in kg/m3 at Different Pressures:
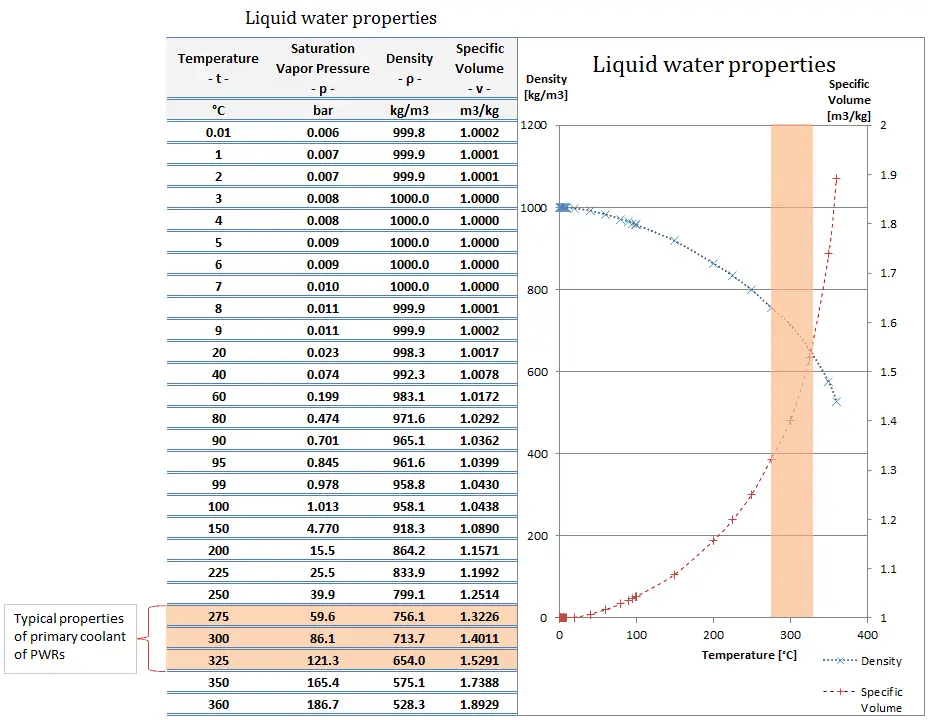
As a result, the mass per unit volume, or density, decreases. This is due to the fact that as water molecules are heated, they gain kinetic energy and move farther apart, occupying a larger volume. The density of water in kg/m3 changes with temperature, with the density decreasing as the temperature increases. Density of Water in kg/m3 at Different Temperatures: Archimedes’ principle is a method that involves measuring the buoyant force of water on an object of known volume, and then using this information to calculate the density of water. Hydrostatic weighing is a technique that involves weighing an object while it is submerged in water, and then using the difference between its weight in air and its weight in water to calculate the density of water. Hydrometry is a method that involves measuring the buoyant force of water on a solid object of known volume and weight, and then using this information to calculate the density of water. The density of water in kg/m3 can be measured using various techniques, including hydrometry, hydrostatic weighing, and Archimedes’ principle. Measurement of Density of Water in kg/m3: Temperature and pressure affect the density of water, with the density increasing as pressure increases and decreasing as temperature increases. Measured using techniques such as hydrometry and hydrostatic weighing, understanding the density of water is crucial in designing underwater structures, managing irrigation systems, and studying aquatic organisms.
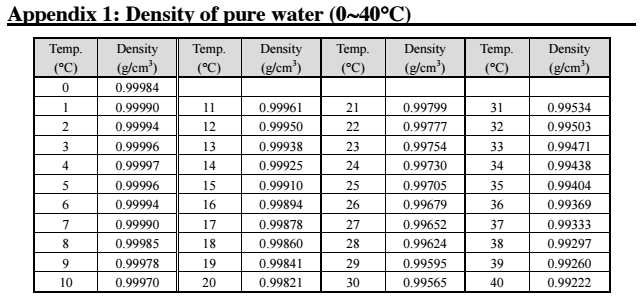
It is important in various fields, including engineering, chemistry, biology, agriculture, and earth sciences. The density of water in kg/m3 is a fundamental physical property that changes with temperature and pressure. This article will explore the density of water in kg/m3, including how it is measured, how it changes with temperature and pressure, and its applications in various fields. In the case of water, the density is typically expressed in kilograms per cubic meter (kg/m3). Density is defined as the amount of mass per unit volume of a substance. The density of water is a fundamental physical property that is important in many fields, including physics, chemistry, engineering, and biology.
